Project Description
alegro
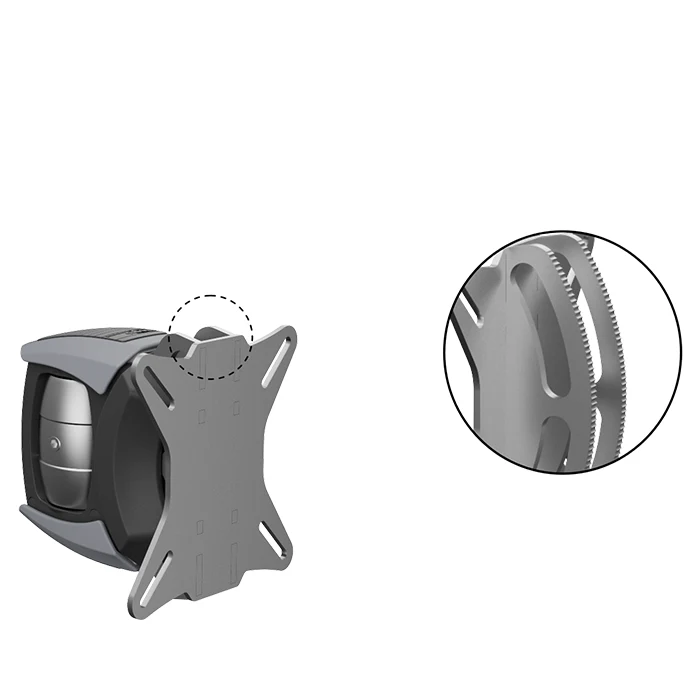
Monitor carrier system
Monitor carrier system
The revolutionizing monitor carrier system allows a single and multiple arrangement of monitors or panel PCs. alegro is characterized by uncomplicated and universal positioning versatility for a vast array of applications.
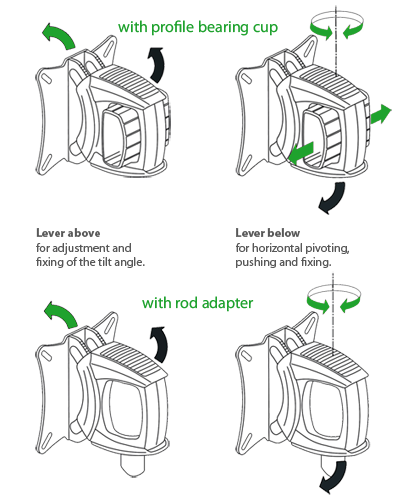
Sturdy construction of the carrier system as a combination of metal, aluminium and plastic. Stable VESA adapter plate of satin-finished stainless steel with slots for VESA standard 75/100. Practical 2-lever mechanism for infinitely variable and precise adjustment of the tilt angle, for horizontal pivoting and fixing to various attachment system parts.
alegro for mounting on the wall, with rod adapter fixed in the carrier profiles of the cart systems fuego, toro, swingo or keo. Otherwise with profile bearing cup for adaption on a horizontal arm, on a swivel arm, on a articulated arm or double articulated arm.
In medicine, industry and all technical fields.
| Load capacity:: | up to 12 kg / 18 kg |
| VESA plate: | satin-finished stainless steel for VESA standard 75/100 |
| Carrier system: | made of metal, aluminium, plastic |
| Adjustment levers: | high-quality, sturdy plastic |
| For more detailed information on the technical details and materials please refer to the type list. | |
- Central carrier pivot with profile bearing cup for aluminium traverse or with rod adapter for round rod.
- Integrated spring locking for optimum, precise adjustment of the monitor.
- Horizontally and vertically rotatable, tiltable and pivotable.
- Secure, precise and simple fixing of the monitor position.
- Synchronous adjustment for double monitoring.
- Optimum cable duct with cable clip externally on the profile or with cable inlet and outlet inside the profile.
- High loading capacity up to 12 kg or 18 kg. 23 kg and special mounting possibilities on request.
- Design and form harmoniously matched to the HAEBERLE cart systems.
This certifies the audit company DQS GmbH according to the certificate registration no. 524821 MP2016.
The self-declaration certificate DIN EN ISO 9001:2015 declares that a quality management system has been introduced and is continuously applied:
SD DIN EN ISO 9001:2015
The certificate DIN EN ISO 13485:2016 approves that the quality management is allowed for the production of medical products:
DIN EN ISO 13485