Project Description
doppio
Equipment cart system · heavy duty
Equipment cart system · heavy duty
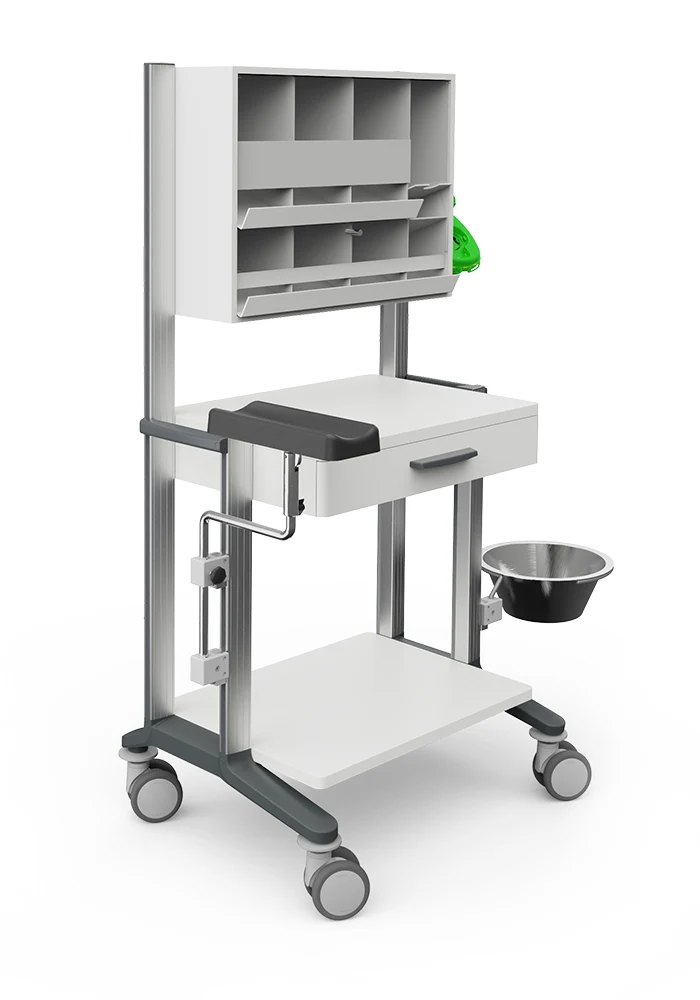
The multifunctional equipment cart system stand out with its modern, puristical appearance. Its sophisticated design with elegant aluminium carrier profiles and high-quality shelves made of wood/plastic blend aesthetically into a modern clinic and practice ambience.
The system widths of 45 cm an 60 cm offer sufficient space to meet all requirements and the large selection of system parts and accessories allow plenty of possibilities for individual design.
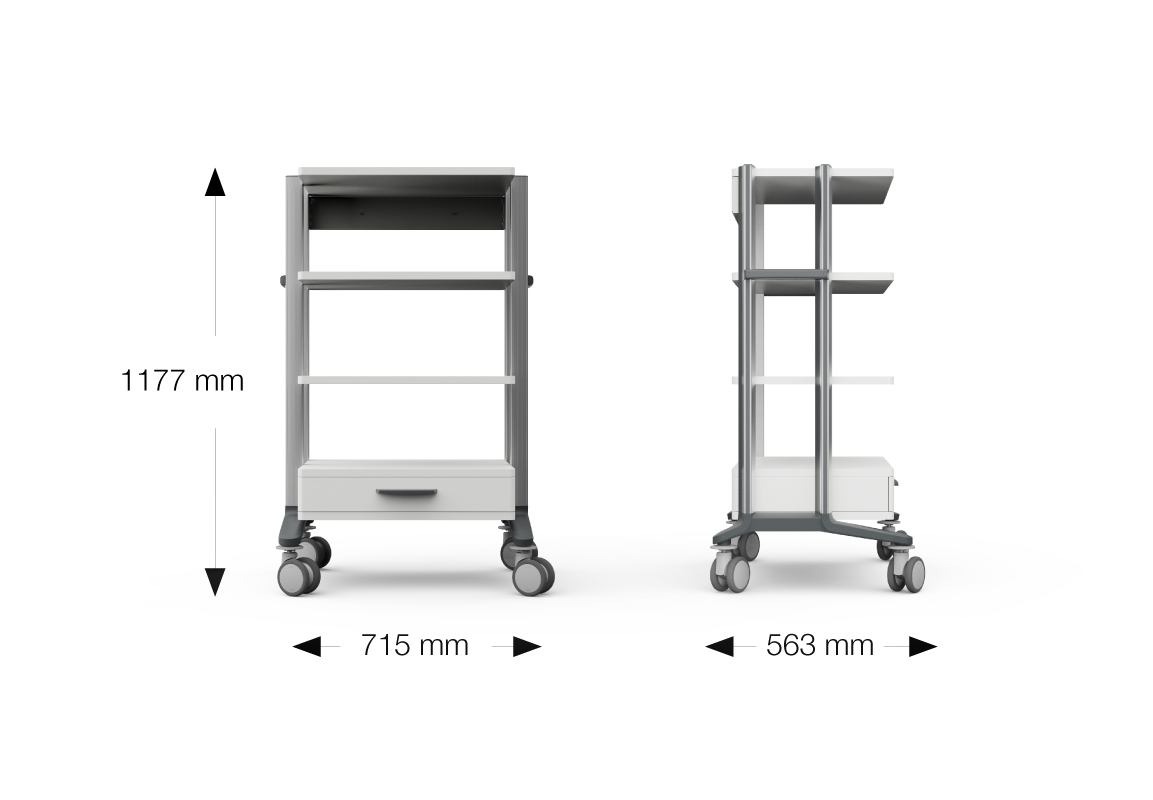
| Cart outer dimensions: | Width: 565 / 715 mm Depth: 571 mm Height: 793 / 834 / 1177 / 1433 mm (Castors with Ø 100 mm) |
| Shelf outer dimensions: | for cart 45 450 x 350 / 450 mm (w x d) for cart 60 600 x 350 / 450 mm (w x d) |
| Shelf load limit: | up to 30 kg |
| Drawer surface load: | up to 10 kg |
| Total load capacity: | up to 120 kg |
| Planning guide for doppio | |
| For more detailed information on the technical details and materials, please refer to the type list. | |
- Sturdy chassis with high-quality, light-running castors and wall guards. Electrically conductive castors optionally for an extra charge.
- 4 puristical aluminium carrier profiles with profile cover caps and integrated cable ducts for convenient cable management.
- Decorative strips in the profiles in grey or blue available.
- Loadable, white-coated shelves made of wood/plastic with rounded edges.
- Beautifully shaped drawers with rounded edges, grey drawer handle and easy-sliding ball telescopic guide with snap-in mechanism.
- Handy side push handles in grey.
This certifies the audit company DQS GmbH according to the certificate registration no. 524821 MP2016.
The self-declaration certificate DIN EN ISO 9001:2015 declares that a quality management system has been introduced and is continuously applied:
SD DIN EN ISO 9001:2015
The certificate DIN EN ISO 13485:2016 approves that the quality management is allowed for the production of medical products:
DIN EN ISO 13485